Stratospheric Ozone Depletion (9.1) Reducing Ozone Depletion (9.2)
Read the text and watch the videos linked below.
Stratospheric Ozone Depletion
We have seen that tropospheric or ground-level pollution contributes to a number of problems in the natural world, exacerbates asthma and breathing difficulties in humans and contributes to the incidence of cancer. Now we turn to the effects of certain pollutants in the stratosphere.
In the troposphere, ozone is an oxidant that can harm respiratory systems in animals and damage a number of structures in plants. However, in the stratosphere ozone forms a necessary, protective shield against radiation from the Sun; it absorbs ultraviolet light and prevents harmful radiation from reaching Earth.
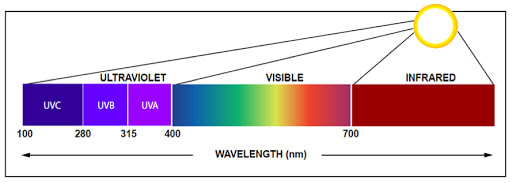
The sun radiates energy at many different wavelengths, including the ultraviolet (UV) waves. The UV waves are further classified into UVA, or low-energy ultraviolet radiation, and the shorter, higher-energy UVB and UVC wavelengths. UV radiation of all types can damage the tissues and DNA of living organisms. Exposure to UVB radiation increases the risk of skin cancer, cataracts and other eye problems, and suppresses the immune system in humans. Exposure to UVB is also harmful to the cells of plants, and it reduces their ability to convert sunlight into usable energy. UVB exposure can therefore harm entire biological communities. For example, losses of phytoplankton--the microscopic algae that form the base of many marine food chains--will cause depletion of fisheries.
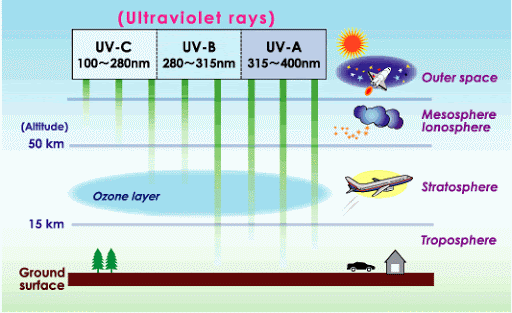
A layer of ozone in the stratosphere absorbs ultraviolet radiation, filtering out harmful UV rays from the Sun. Its ability to to absorb UV radiation and thereby shield the surface below makes stratospheric ozone critically important to life on Earth.
- The stratospheric ozone layer is important to the evolution of life on Earth and the continued health and survival of life on Earth.
- A decrease in stratospheric ozone increases the UV rays that reach the Earth's surface. Exposure to UV rays can lead to skin cancer and cataracts in humans.
We have seen that tropospheric or ground-level pollution contributes to a number of problems in the natural world, exacerbates asthma and breathing difficulties in humans and contributes to the incidence of cancer. Now we turn to the effects of certain pollutants in the stratosphere.
In the troposphere, ozone is an oxidant that can harm respiratory systems in animals and damage a number of structures in plants. However, in the stratosphere ozone forms a necessary, protective shield against radiation from the Sun; it absorbs ultraviolet light and prevents harmful radiation from reaching Earth.
The sun radiates energy at many different wavelengths, including the ultraviolet (UV) waves. The UV waves are further classified into UVA, or low-energy ultraviolet radiation, and the shorter, higher-energy UVB and UVC wavelengths. UV radiation of all types can damage the tissues and DNA of living organisms. Exposure to UVB radiation increases the risk of skin cancer, cataracts and other eye problems, and suppresses the immune system in humans. Exposure to UVB is also harmful to the cells of plants, and it reduces their ability to convert sunlight into usable energy. UVB exposure can therefore harm entire biological communities. For example, losses of phytoplankton--the microscopic algae that form the base of many marine food chains--will cause depletion of fisheries.
A layer of ozone in the stratosphere absorbs ultraviolet radiation, filtering out harmful UV rays from the Sun. Its ability to to absorb UV radiation and thereby shield the surface below makes stratospheric ozone critically important to life on Earth.
Formation of Stratospheric Ozone
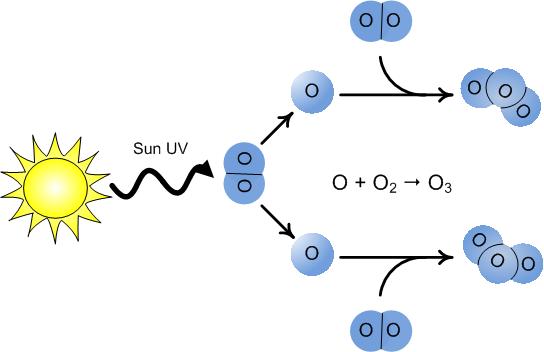
When solar radiation strikes O2 in the stratosphere, a series of chemical reactions begins that produces a new molecule: ozone (O3). In the first step, UVC radiation breaks the molecular bond holding an oxygen molecule together:
O2 + UVC → O + O
This happens to only a few oxygen molecules at any given time. The vast majority of the oxygen in the atmosphere remains in to form O2. In the second step, a free oxygen atom (O) produced in the first reaction encounters an oxygen molecule and they form ozone:
O + O2 → O3
Both UVB and UVC radiation can break a bond in this new ozone molecule, forming molecular oxygen and a free oxygen atom once again:
O3 + UVB or UVC → O2 + O
Thus formation of ozone in the presence of sunlight and its subsequent breakdown is a cycle that can occur indefinitely as long as there is UV energy entering the atmosphere. Under normal conditions, the amount of ozone in the stratosphere remains at a steady state.
When solar radiation strikes O2 in the stratosphere, a series of chemical reactions begins that produces a new molecule: ozone (O3). In the first step, UVC radiation breaks the molecular bond holding an oxygen molecule together:
O2 + UVC → O + O
This happens to only a few oxygen molecules at any given time. The vast majority of the oxygen in the atmosphere remains in to form O2. In the second step, a free oxygen atom (O) produced in the first reaction encounters an oxygen molecule and they form ozone:
O + O2 → O3
Both UVB and UVC radiation can break a bond in this new ozone molecule, forming molecular oxygen and a free oxygen atom once again:
O3 + UVB or UVC → O2 + O
Thus formation of ozone in the presence of sunlight and its subsequent breakdown is a cycle that can occur indefinitely as long as there is UV energy entering the atmosphere. Under normal conditions, the amount of ozone in the stratosphere remains at a steady state.
|
The video at the link below is older, but does a great job of illustrating the chemistry behind ozone formation and depletion.
|
Depletion of Stratospheric Ozone
For many years, the same chemicals that made refrigeration and air conditioning possible were also used in a host of other consumer items, including aerosol spray cans and products such as Styrofoam. These chemicals, called chlorofluorocarbons, or CFCs, were considered essential to modern life, and producing them was a multibillion-dollar industry. CFCs were considered "safe" because they are both nontoxic and nonflammable. But it turned out that these chemicals had adverse effects by promoting the breakdown of ozone in the stratosphere.
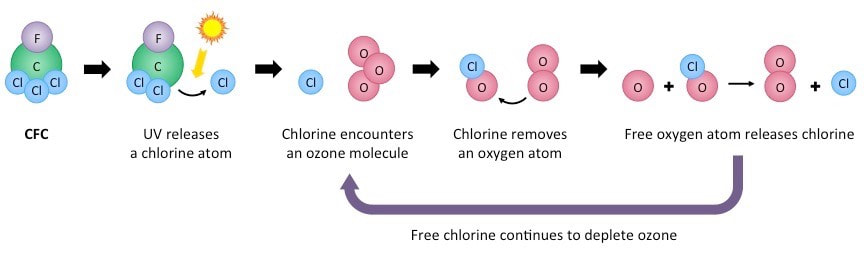
CFCs introduce chlorine (Cl) into the stratosphere. When chlorine is present, it can attach to an oxygen atom in an ozone molecule, thereby breaking the bond between that atom and the molecule and forming chlorine monoxide (ClO) and O2.
O3 + Cl → ClO + O2
Subsequently, the chlorine monoxide molecule reacts with a free oxygen atom, which pulls the oxygen from the ClO to produce free chlorine again.
ClO + O → Cl + O2
When we consider these reactions together, we see that chlorign starts out and ends up as a free Cl atom. In contrast, an ozone molecule and a free oxygen atom are converted into two oxygen molecules. A substance that aids a reaction but does not get used up itself is called a catalyst. A single chlorine atom can catalyze the breakdown of as many as 100,000 ozone molecules until finally one chlorine atom finds another an the process is stopped. In the process, the ozone molecules are no longer available to absorb incoming UVB radiation. As a result, the UVB radiation can reach Earth's surface and cause harm to biological organisms.
- Stratospheric ozone depletion is caused by anthropogenic factors, such as chlorofluorocarbons (CFCs), and natural factors, such as the melting of ice crystals in the atmosphere at the beginning of the Antarctic spring
For many years, the same chemicals that made refrigeration and air conditioning possible were also used in a host of other consumer items, including aerosol spray cans and products such as Styrofoam. These chemicals, called chlorofluorocarbons, or CFCs, were considered essential to modern life, and producing them was a multibillion-dollar industry. CFCs were considered "safe" because they are both nontoxic and nonflammable. But it turned out that these chemicals had adverse effects by promoting the breakdown of ozone in the stratosphere.
CFCs introduce chlorine (Cl) into the stratosphere. When chlorine is present, it can attach to an oxygen atom in an ozone molecule, thereby breaking the bond between that atom and the molecule and forming chlorine monoxide (ClO) and O2.
O3 + Cl → ClO + O2
Subsequently, the chlorine monoxide molecule reacts with a free oxygen atom, which pulls the oxygen from the ClO to produce free chlorine again.
ClO + O → Cl + O2
When we consider these reactions together, we see that chlorign starts out and ends up as a free Cl atom. In contrast, an ozone molecule and a free oxygen atom are converted into two oxygen molecules. A substance that aids a reaction but does not get used up itself is called a catalyst. A single chlorine atom can catalyze the breakdown of as many as 100,000 ozone molecules until finally one chlorine atom finds another an the process is stopped. In the process, the ozone molecules are no longer available to absorb incoming UVB radiation. As a result, the UVB radiation can reach Earth's surface and cause harm to biological organisms.
In the mid-1980s, atmospheric researchers notices that stratospheric ozone in Antarctica had been decreasing each year, beginning in about 1979. Since the late 1970s, global ozone concentrations had decreased by more than 10%. Depletion was greatest at the poles, but occurred worldwide. Researchers also determined that, in the Antarctic, ozone depletion was seasonal: Each year the depletion occurred from roughly August through November (late winter through early spring in the Southern Hemisphere). The depletion caused an area of severely reduced ozone concentrations over most of Antarctica, creating what has come to be called the "ozone hole."
The cause of the formation of the ozone hole is complex. It appears that extremely cold weather conditions during the polar winter cause a buildup of ice crystals mixed with nitrogen dioxide. This in turn provides the perfect surface for the formation of the stable molecule Cl2, which accumulates as atmospheric chlorine interacts with the ice crystals. When the Sun reappears in the spring, UV radiation breaks down this molecule into Cl again, which in turn catalyzes the destruction of ozone as described above. Because almost no ozone forms in the dark of the polar winter, a large amount of thinning occurs. Only after the temperatures warm up and the chlorine gets diluted by air coming from outside the polar region does the thinning diminish. Decreased stratospheric ozone has led to a rise in the amount of UVB radiation that reaches the surface of Earth.
The cause of the formation of the ozone hole is complex. It appears that extremely cold weather conditions during the polar winter cause a buildup of ice crystals mixed with nitrogen dioxide. This in turn provides the perfect surface for the formation of the stable molecule Cl2, which accumulates as atmospheric chlorine interacts with the ice crystals. When the Sun reappears in the spring, UV radiation breaks down this molecule into Cl again, which in turn catalyzes the destruction of ozone as described above. Because almost no ozone forms in the dark of the polar winter, a large amount of thinning occurs. Only after the temperatures warm up and the chlorine gets diluted by air coming from outside the polar region does the thinning diminish. Decreased stratospheric ozone has led to a rise in the amount of UVB radiation that reaches the surface of Earth.
Reducing Ozone Depletion
In response to the decrease in stratospheric ozone, 24 nations in 1987 signed the Montreal Protocol on Substances that Deplete the Ozone Layer. This was a commitment to reduce CFC production by 50% by the year 2000. It was the most far-reaching environmental treaty to date, in which global CFC exporters like the U.S. appeared in some ways to prioritize the protection of the global biosphere over their short-term economic self-interest. More than 180 countries eventually signed a series of increasingly stringent amendments that required the elimination of CFC production and use in the developed world by 1996. In total, the protocol addressed 96 ozone-depleting compounds.
Because of these effort, the concentration of chlorine in the stratosphere peaked at about 4 ppb and is now decreasing. The chlorine concentration reduction process is slow because CFCs are not easily removed from the stratosphere. With the stabilization of chlorine concentrations, stratospheric ozone depletion should decrease in subsequent decades.
Unfortunately, many of the alternative refrigerants that are less harmful to the ozone layer, including a group of gases known as hydrochlorofluorocarbons (HCFCs), still have very high greenhouse warming potentials. As a result, all countries will phase out the use of HCFCs by 2030.
- Ozone depletion can be mitigated by replacing ozone-depleting chemicals with substitutes that do not deplete the ozone layer. Hydrofluorocarbons (HFCs) are one such replacement, but some are strong greenhouse gases.
In response to the decrease in stratospheric ozone, 24 nations in 1987 signed the Montreal Protocol on Substances that Deplete the Ozone Layer. This was a commitment to reduce CFC production by 50% by the year 2000. It was the most far-reaching environmental treaty to date, in which global CFC exporters like the U.S. appeared in some ways to prioritize the protection of the global biosphere over their short-term economic self-interest. More than 180 countries eventually signed a series of increasingly stringent amendments that required the elimination of CFC production and use in the developed world by 1996. In total, the protocol addressed 96 ozone-depleting compounds.
Because of these effort, the concentration of chlorine in the stratosphere peaked at about 4 ppb and is now decreasing. The chlorine concentration reduction process is slow because CFCs are not easily removed from the stratosphere. With the stabilization of chlorine concentrations, stratospheric ozone depletion should decrease in subsequent decades.
Unfortunately, many of the alternative refrigerants that are less harmful to the ozone layer, including a group of gases known as hydrochlorofluorocarbons (HCFCs), still have very high greenhouse warming potentials. As a result, all countries will phase out the use of HCFCs by 2030.