Reduction of Air Pollutants
- Methods to reduce air pollutants include regulatory practices, conservation practices, and alternative fuels.
- A vapor recovery nozzle is an air pollution control device on a gasoline pump that prevents fumes from escaping into the atmosphere when fueling a motor vehicle
- A catalytic converter is an air pollution control device for internal combustion engines that converts pollutants (CO, NOx and hydrocarbons) in exhaust into less harmful molecules (CO2, N2, O2 and H2O)
- Wet and dry scrubbers are air pollution control devices that remove particulates and/or gases from industrial exhaust systems
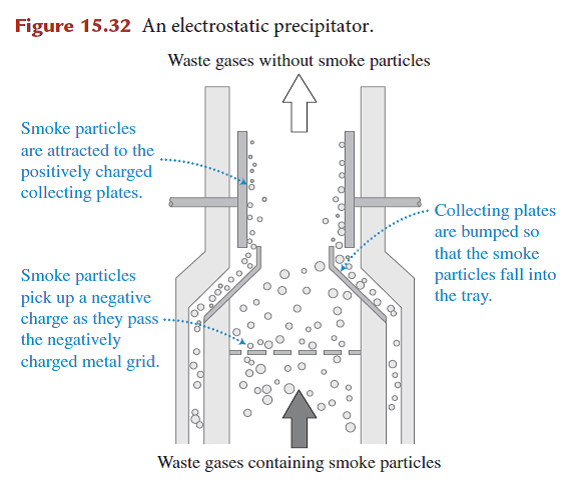
- Methods to reduce air pollution from coal-burning power plants include scrubbers and electrostatic precipitators
Wet Scrubbers
The polluted gas stream is brought into contact with the scrubbing liquid, by spraying it with the liquid or by forcing it through a pool of liquid. Wet Scrubbers remove pollutant gases by dissolving or absorbing them into the liquid.
The polluted gas stream is brought into contact with the scrubbing liquid, by spraying it with the liquid or by forcing it through a pool of liquid. Wet Scrubbers remove pollutant gases by dissolving or absorbing them into the liquid.

Advantages
Disadvantages
- No secondary dust sources
- Small space requirements
- Ability to collect gases as well as particulates
- Ability to handle high-temperature, high-humidity gas streams
- Capital cost low (if waste water treatment system not required)
- Minimal fire and explosion hazards
- Ability to achieve high collection efficiency (>95%)
Disadvantages
- Creation of water-disposal problem
- Waste product collected wet
- Solids buildup at the wet-dry interface
- Relatively high maintenance costs
- Must be protected from freezing
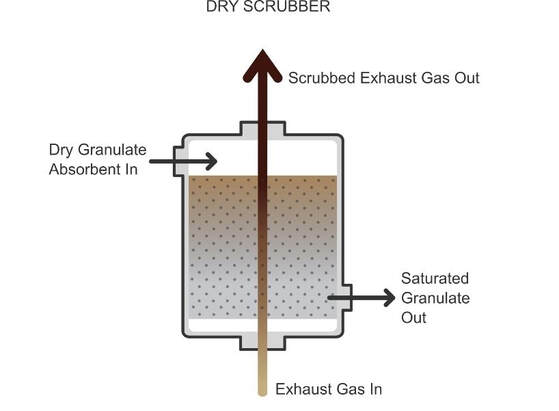
Dry Scrubbers
Dry scrubbers spray very fine absorbents into a vessel where they combine with flue gases or the flue gases are passed over granulate absorbent packing bed. The absorbent chemically reacts with the sulfur dioxide in the flue gases containing sulfur dioxide, removing the sulfur dioxide from the flue gases. The spent absorbent must then be removed and replaced with fresh absorbent.
Dry scrubbers spray very fine absorbents into a vessel where they combine with flue gases or the flue gases are passed over granulate absorbent packing bed. The absorbent chemically reacts with the sulfur dioxide in the flue gases containing sulfur dioxide, removing the sulfur dioxide from the flue gases. The spent absorbent must then be removed and replaced with fresh absorbent.
Advantages
A dry or semi-dry scrubbing system, unlike wet scrubbers, does not saturate the flue gas stream that is being treated with liquid. In some cases no liquid is added, while in others only the amount of liquid that can be evaporated in the flue gas without condensing is added. Therefore, dry scrubbers generally do not have waste water handling/disposal requirements.
Disadvantages
A dry or semi-dry scrubbing system, unlike wet scrubbers, does not saturate the flue gas stream that is being treated with liquid. In some cases no liquid is added, while in others only the amount of liquid that can be evaporated in the flue gas without condensing is added. Therefore, dry scrubbers generally do not have waste water handling/disposal requirements.
Disadvantages
- Lower collection efficiency than wet scrubber (<95%).
- Dry scrubbers typically use lime or limestone that have much slower reaction rates than sodium hydroxide, potassium hydroxide, and magnesium hydroxide utilized in web scrubbers. The result is an increase in scrubber size when compared to wet scrubbers.
- Disposal costs for spent absorbent
Noise Pollution
- Noise pollution is sound at levels high enough to cause physiological stress and hearing loss
- Sources of noise pollution in urban areas include transportation, construction, and domestic and industrial activity
- Some effects of noise pollution on animals in ecological systems include stress, the masking of sounds used to communicate or hunt, damaged hearing, and causing changes to migratory routes
Acid Rain
- Acid Rain and deposition is due to nitrogen oxides and sulfur oxides from anthropogenic and natural sources in the atmosphere
- Nitric oxides that cause acid deposition come from motor vehicles and coal-burning power plants
- Sulfur dioxides that cause acid deposition come from coal-burning power plants
- Acid deposition mainly affects communities that are downwind from coal-burning power plants
- Acid rain and deposition can lead to the acidification of soils and bodies of water and corrosion of human-made structures
- Regional differences in soils and bedrock affect the impact that acid deposition has on the region--such as limestone bedrock's ability to neutralize the effect of acid rain on lakes and ponds
Acid Deposition
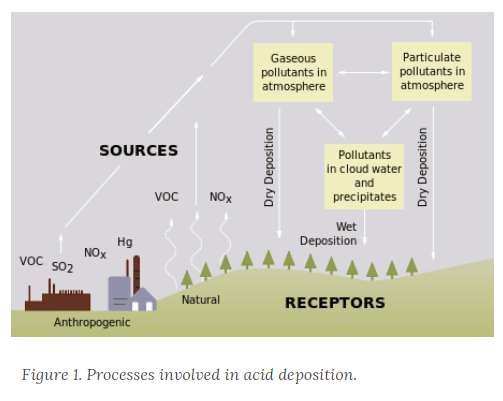
Acid rain is a term referring to a mixture of wet and dry deposition (deposited material) from the atmosphere containing higher than normal amounts of nitric and sulfuric acids. The precursors, or chemical forerunners, of acid rain formation result from both natural sources, such as volcanoes and decaying vegetation, and man-made sources, primarily emissions of sulfur dioxide (SO2) and nitrogen oxides (NOx) resulting from fossil fuel combustion. Acid rain occurs when these gases react in the atmosphere with water, oxygen, and other chemicals to form various acidic compounds. The result is a mild solution of sulfuric acid and nitric acid. When sulfur dioxide and nitrogen oxides are released from power plants and other sources, prevailing winds blow these compounds across state and national borders, sometimes over hundreds of miles.
Measuring Acid Rain
Acid rain is measured using a scale called “pH.” The lower a substance’s pH, the more acidic it is. Pure water has a pH of 7.0. However, normal rain is slightly acidic because carbon dioxide (CO2) dissolves into it forming weak carbonic acid, giving the resulting mixture a pH of approximately 5.6 at typical atmospheric concentrations of CO2. As of 2000, the most acidic rain falling in the U.S. has a pH of about 4.3.
Effects of Acid Rain
Acid rain causes acidification of lakes and streams and contributes to the damage of trees at high elevations (for example, red spruce trees above 2,000 feet) and many sensitive forest soils. In addition, acid rain accelerates the decay of building materials and paints, including irreplaceable buildings, statues, and sculptures that are part of our nation’s cultural heritage. Prior to falling to the earth, sulfur dioxide (SO2) and nitrogen oxide (NOx) gases and their particulate matter derivatives—sulfates and nitrates—contribute to visibility degradation and harm public health.
The ecological effects of acid rain are most clearly seen in the aquatic, or water, environments, such as streams, lakes, and marshes. Most lakes and streams have a pH between 6 and 8, although some lakes are naturally acidic even without the effects of acid rain. Acid rain primarily affects sensitive bodies of water, which are located in watersheds whose soils have a limited ability to neutralize acidic compounds (called “buffering capacity”). Lakes and streams become acidic (i.e., the pH value goes down) when the water itself and its surrounding soil cannot buffer the acid rain enough to neutralize it. In areas where buffering capacity is low, acid rain releases aluminum from soils into lakes and streams; aluminum is highly toxic to many species of aquatic organisms. Acid rain causes slower growth, injury, or death of forests. Of course, acid rain is not the only cause of such conditions. Other factors contribute to the overall stress of these areas, including air pollutants, insects, disease, drought, or very cold weather. In most cases, in fact, the impacts of acid rain on trees are due to the combined effects of acid rain and these other environmental stressors. Acid rain and the dry deposition of acidic particles contribute to the corrosion of metals(such as bronze) and the deterioration of paint and stone (such as marble and limestone). These effects significantly reduce the societal value of buildings, bridges, cultural objects (such as statues, monuments, and tombstones), and cars (Figure below).
Measuring Acid Rain
Acid rain is measured using a scale called “pH.” The lower a substance’s pH, the more acidic it is. Pure water has a pH of 7.0. However, normal rain is slightly acidic because carbon dioxide (CO2) dissolves into it forming weak carbonic acid, giving the resulting mixture a pH of approximately 5.6 at typical atmospheric concentrations of CO2. As of 2000, the most acidic rain falling in the U.S. has a pH of about 4.3.
Effects of Acid Rain
Acid rain causes acidification of lakes and streams and contributes to the damage of trees at high elevations (for example, red spruce trees above 2,000 feet) and many sensitive forest soils. In addition, acid rain accelerates the decay of building materials and paints, including irreplaceable buildings, statues, and sculptures that are part of our nation’s cultural heritage. Prior to falling to the earth, sulfur dioxide (SO2) and nitrogen oxide (NOx) gases and their particulate matter derivatives—sulfates and nitrates—contribute to visibility degradation and harm public health.
The ecological effects of acid rain are most clearly seen in the aquatic, or water, environments, such as streams, lakes, and marshes. Most lakes and streams have a pH between 6 and 8, although some lakes are naturally acidic even without the effects of acid rain. Acid rain primarily affects sensitive bodies of water, which are located in watersheds whose soils have a limited ability to neutralize acidic compounds (called “buffering capacity”). Lakes and streams become acidic (i.e., the pH value goes down) when the water itself and its surrounding soil cannot buffer the acid rain enough to neutralize it. In areas where buffering capacity is low, acid rain releases aluminum from soils into lakes and streams; aluminum is highly toxic to many species of aquatic organisms. Acid rain causes slower growth, injury, or death of forests. Of course, acid rain is not the only cause of such conditions. Other factors contribute to the overall stress of these areas, including air pollutants, insects, disease, drought, or very cold weather. In most cases, in fact, the impacts of acid rain on trees are due to the combined effects of acid rain and these other environmental stressors. Acid rain and the dry deposition of acidic particles contribute to the corrosion of metals(such as bronze) and the deterioration of paint and stone (such as marble and limestone). These effects significantly reduce the societal value of buildings, bridges, cultural objects (such as statues, monuments, and tombstones), and cars (Figure below).
Sulfates and nitrates that form in the atmosphere from sulfur dioxide (SO2) and nitrogen oxides (NOx) emissions contribute to visibility impairment, meaning we cannot see as far or as clearly through the air. The pollutants that cause acid rain—sulfur dioxide (SO2) and nitrogen oxides (NOx)—damage human health. These gases interact in the atmosphere to form fine sulfate and nitrate particles that can be transported long distances by winds and inhaled deep into people’s lungs. Fine particles can also penetrate indoors. Many scientific studies have identified a relationship between elevated levels of fine particles and increased illness and premature death from heart and lung disorders, such as asthma and bronchitis.